The corona vaccine & logistics
Last Wednesday on November 18th, the Mainz-based BioNTech Group, together with Pfizer, announced the results of the phase 3 trial of its corona vaccine. The results indicate that the vaccine offers a good 95% protection against COVID-19 - about 5% more than was suspected in phase 2. This is a groundbreaking development, as the vaccine is the first in the world to achieve success in the final phase of the study. Due to the still steeply increasing numbers of infections in the U.S., BioNTech and Pfizer intend to apply for emergency approval for the use of the vaccine by the end of November, so that the first vaccinations can already be carried out in December. BioNTech and Pfizer also plan to produce around 50 million doses of the vaccine by the end of 2020. In 2021, up to 1.3 billion doses are to be expected.
These prospects present the logistics industry in particular with the challenge of handling and organizing the distribution of the enormous number of individual doses of the vaccine within a short period of time.
Warehousing1 has therefore compiled a list of the factors that German and international logistics companies must take into account when handling logistics for the corona vaccine.
What needs to be considered in logistics regarding the distribution of the COVID-19 vaccine
- The correct cooling
Cooling is an essential factor in the distribution of the SARS-CoV-2 agent. At the present time, cooling of the cans at -70°C is recommended and planned. In their press release, BioNtech and Pfizer themselves talk about handling the logistics using their existing cold chain infrastructure.
They have developed special temperature-stable shipping units that can cool the vaccine with dry ice for up to 15 days. A special GPS temperature sensor in each shipping unit will ensure that the cold chain along the supply route is not interrupted at any time. In an interview with Reuters , BioNTech boss Ugur Sahin said that the vaccine could also be stored in the refrigerator for up to five days. However, further tests are currently being conducted to determine the actual storage period and other specifics.
In a Study. with McKinsey, DHL cited three archetypal models that could also be used for the logistics to the end customer. The following image illustrates this clearly:
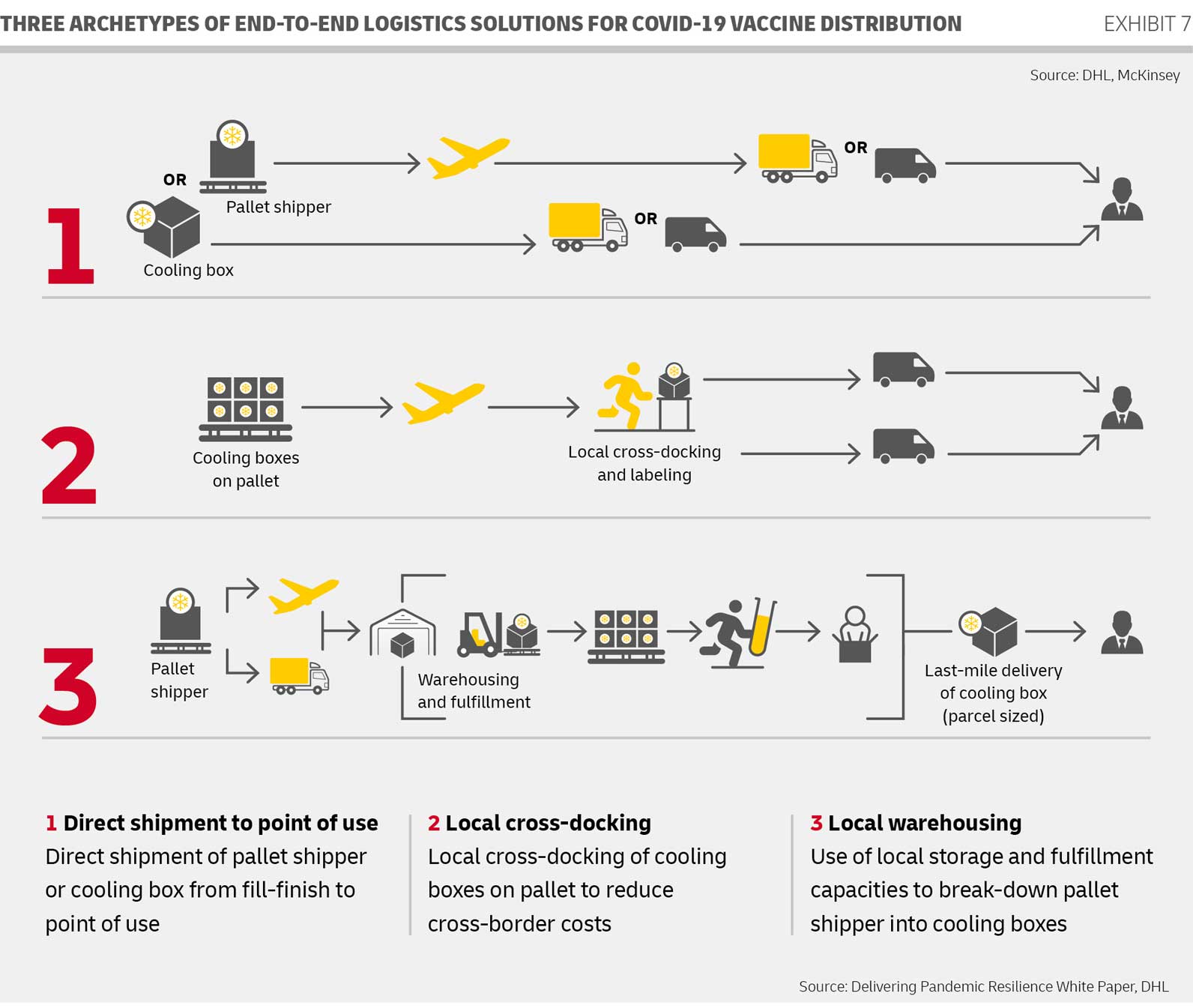
It is clear that there cannot and will not be one single correct distribution method. Whether local warehousing and Fulfillment as in step 3 or local Dropshipping is used, short delivery routes without many stops are advantageous, especially because of the necessary assurance of -70°C.
- Networks must be extended and adapted
So why is the distribution of the corona vaccine such a challenge for the logistics industry? The main reason is that pharmaceutical logistics not only need to improve the transport itself, but also require a new strategy for individual distribution networks.
Especially in Germany, it is to be expected that Frankfurt Airport will play a decisive role in the transportation of corona vaccines. The airport operator Fraport already has 20 thermal transportersthat can be used to transport temperature-sensitive pharmaceuticals.
The Swiss logistics giant Kühne+Nagel has also expanded its logistics network to include temperature-controlled facilities for air traffic in Brussels and Johannesburg. These new locations are intended to facilitate and strengthen the distribution of temperature-sensitive products such as the corona vaccine.
According to an article in the Swiss magazine Blick (22.11.20), the vaccine is to be stored and transshipped in Aargau in Switzerland - in the small town of Möhlin, not far from the German border. In recent years, the small town has turned into an important logistical hub in Switzerland; above all, the proximity to the airports of Zurich and Basel Mulhouse Freiburg was a decisive factor in the choice of this location as a storage site for the COVID 19 vaccine.
- The potential of micro-fulfillment
In general, innovative and complex processes are required for pharmaceutical logistics. The so-called micro-fulfillment would also be of great advantage in the case of transport of the corona vaccine. With this model, goods move closer to the end consumer, which means that the final delivery can be handled a lot faster. The goods are brought from supraregional depots, for example outside the city, to the so-called micro-depots or hubs.
From the micro-distribution centers, the goods - or, in this case, the corona vaccine - can be brought directly to the delivery location. This results in a more reliable and faster delivery, which is particularly necessary in the medical sector.
Conclusion
How the distribution of the COVID-19 vaccine will be handled in detail will only become clear when the time comes. In the meantime, logistics entrepreneurs should work on expanding and improving their networks to guarantee efficient delivery routes and strategies. It is important to be prepared for all eventualities - such as the pandemic itself is and was one.
If you yourself see a need for improvement in your logistics setup, Warehousing1 is the perfect partner. Our account managers will be happy to explain to you in a non-binding consultation which possibilities there are for you in a cooperation with Warehousing1 and one of our more than 500 partner locations. Please contact us directly via our contact form to learn more about the services of Warehousing1.